This post is a continuation of Part I: vs s: what are they and what’s the difference?
Progestins are synthetic forms of the sex , , that are used in . The s in help prevent an egg from being released or implanted in your , and they are highly effective at doing so. There are different kinds, or generations, of s, and they can have slightly different side effects depending on what kind they are.
Most varieties of are derived from other hormones, namely or . Because of this, s have a very similar chemical structure to these other hormones, and can sometimes have unintentional effects on those systems in the body. Meanwhile, when s break down in the body, the resulting products can also interact with other pathways, like those related to s or s. Throughout this article, we’ll explain which s have these so-called “androgenic effects” or “estrogenic effects” and how they can inform which type of is the best choice for your .
What are generations?
Progestins are often grouped in terms of “generations.” This simply refers to when the was first available on the market. Notably, it doesn’t mean all s of a certain generation are the same.
That said, earlier-generation s overall tend to have more androgenic activity, meaning they impact the pathway to a greater extent.1 s are often thought of as ‘male hormones,’ though actually everyone has them. But they are produced in much higher amounts by testes than by .2 This androgenic activity can change what kind of side effects will have, and what you might experience when using it.
Like what you’re reading? Get the latest straight to your inbox 💌
Commonly prescribed s
Some commonly prescribed s and their generations are described below, as well as the degree to which each regulates other steroid pathways, and thus may lead to additional side effects.
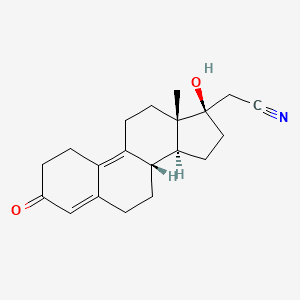
Norethindrone
Norethindrone is a first-generation that is commonly found in oral contraceptives like the pill. It first entered the market for this use in 1962.3 Norethindrone is available in both combined oral contraceptives and progestin-only forms of .
In addition to having low-to-moderate effects in its role as a , norethindrone also has properties that cause it to impact and systems in the body. The estrogenic effects are due in part to the fact that one of norethindrone’s breakdown products (metabolites) is , a form of .4
These estrogenic effects are mild. Norethindrone’s effects at both the and receptors are not as strong as some of the subsequently developed s.
The chemical structure of norethindrone. (Credit: PubChem)
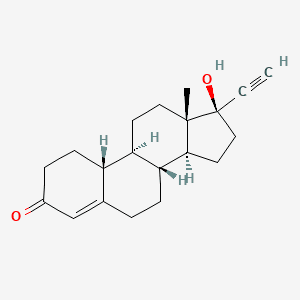
Levonorgestrel
Levonorgestrel is a second-generation and can be found in many brands of combined oral contraceptives, emergency contraceptives, the mini pill, and the hormonal IUD. It has stronger progestogenic and androgenic effects than norethindrone, so it is more likely to lead to androgenic side effects including acne and worse cholesterol levels which can in theory lead to blood clots.5
However, in a meta-analysis of clinical data, combined oral contraceptives containing levonorgestrel were associated with a lower risk of blood clots than those containing any other .6 So, levonorgestrel’s effects on cholesterol levels don’t seem to result in more blood clots when using the combined pill.
The chemical structure of levonorgestrel. (Credit: PubChem)
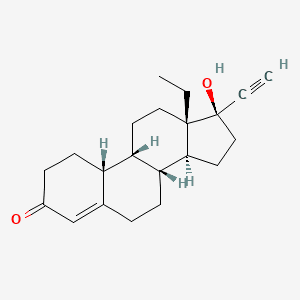
Norgestimate
Norgestimate is a third-generation found in combined pills like Ortho Tri-Cyclen. It has effects similar to , and just slight estrogenic effects. Studies also show that norgestimate can help with acne.7
The chemical structure of norgestimate. (Credit: PubChem)
Dienogest
Dienogest is a fourth-generation with strong progestogenic activity and no androgenic effects. Rather, dienogest has anti-androgenic activity and is not associated with metabolic side effects that can affect things like blood glucose.8 Dienogest is available as a mini pill or in combined oral contraceptives with either or estradiol valerate, and it can be helpful for the treatment of endometriosis.9
The chemical structure of dienogest. (Credit: PubChem)
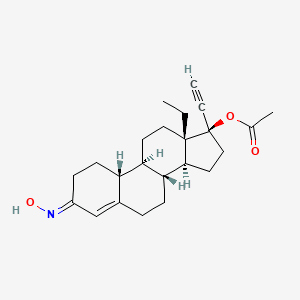
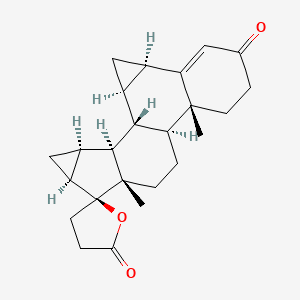
Drosperinone
Drosperinone is more unique among the s: It’s not derived not from or like most other s, but rather from spironolactone, a medication used to block the receptor for the aldosterone.10 If you look at the diagram of drospirenone compared to the others, you (yes, you!) can actually see that it’s an oddball. Drosperinone is therefore structurally different from the other s and is not classified with regard to ‘generation.’
Drospirenone, like its parent molecule spironolactone, decreases the amount of sodium your body retains and modestly reduces blood pressure.11 It can also raise potassium levels, meaning people with kidney disease and some other medical conditions should avoid it.12-13
Drosperinone is available in the mini pill as well as in combined oral contraceptives with and may be used to reduce symptoms of premenstrual dysphoric disorder (PMDD). The risk of blood clots associated with drospirenone is minimally higher than with levonorgestrel, but minimally so, so it doesn’t affect medical recommendations to use it. Drosperinone does not have the negative metabolic effects of levonorgestrel.
The chemical structure of drospirenone. (Credit: PubChem)
Which is right for me?
If you’re interested in using a progestin-only hormonal contraceptive like the IUD or the mini-pill and you’re concerned about acne or metabolic effects, avoiding androgenic s like levonorgestrel and norethindrone might help. These more androgenic s can also cause changes in blood cholesterol levels that could be harmful for your long-term risk of cardiovascular disease. (But note that whether these changes in blood cholesterol levels truly affect cardiovascular risk isn’t yet known.)
When s are delivered with an in combined hormonal contraceptives, the combination therapy acts as an anti-androgen, so these changes tend to be neutralized. Overall, levonorgestrel in combination with a low dose of appears to have the lowest risk of blood clots among the combination oral contraceptives.
That said, if you’re worried about acne or cholesterol levels you may want to avoid the more androgenic s. Additionally, people concerned about body weight gain or high blood sugar levels initially may want to try a like norgestimate or drospirenone.
Importantly, research around uses and side effects of s are based on large studies looking at outcomes and side effects in large cohorts of individuals. Therefore, these data cannot tell us exactly how a certain will affect a certain person, and a that works well for one person may give intolerable side effects for another.
Is adyn right for you? Take the quiz.
Unanswered questions about s
Some important questions about s remain unanswered. More research is needed to understand how different s work in individuals across a wide range of body weights as well as for people with medical conditions such as diabetes or decreased kidney function.
Just as is broken down into metabolites, so are the different s, but these metabolites aren’t always well understood. So although clinical data strongly support the overall safety and effectiveness of s for a number of uses, a great deal of research is still needed to understand the full spectrum of effects of different s and how these effects vary across individuals.
Interested in how using science like this can help you find the best formulation for your body? Check out The Birth Control Test.
-
- Stanczyk, Frank Z. “Structure–function relationships, pharmacokinetics, and potency of orally and parenterally administered Progestogens.” Treatment of the Postmenopausal Woman. Academic Press, 2007. 779-798.
- Hammes, Stephen R., and Ellis R. Levin. “Impact of estrogens in males and androgens in females.” The Journal of Clinical Investigation 129.5 (2019): 1818-1826.
- Butler, Aliya, and Sheraden Seward. “Progestin: Synthetic Progesterone.” Embryo Project Encyclopedia (2008 Nov 18; Updated 2023 Sep 11; Accessed 2024 Mar 20).
- Chu, Micheline C., et al. “Formation of ethinyl estradiol in women during treatment with norethindrone acetate.” The Journal of Clinical Endocrinology & Metabolism 92.6 (2007): 2205-2207.
- Kemmeren, J. M., A. Algra, and D. E. Grobbee. “Effect of second and third generation oral contraceptives on lipid metabolism in the absence or presence of the factor V Leiden mutation.” Journal of Internal Medicine 250.5 (2001): 441-448.
- Oedingen, Carina, Stefan Scholz, and Oliver Razum. “Systematic review and meta-analysis of the association of combined oral contraceptives on the risk of venous thromboembolism: the role of the progestogen type and estrogen dose.” Thrombosis Research 165 (2018): 68-78.
- Lucky, Anne W., et al. “Effectiveness of norgestimate and ethinyl estradiol in treating moderate acne vulgaris.” Journal of the American Academy of Dermatology 37.5 (1997): 746-754.
- Regidor, Pedro-Antonio, and Adolf E. Schindler. “Antiandrogenic and antimineralocorticoid health benefits of COC containing newer progestogens: dienogest and drospirenone.” Oncotarget 8.47 (2017): 83334.
- Schindler, Adolf E. “Dienogest in long-term treatment of endometriosis.“ International Journal of Women’s Health (2011): 175-184.
- Cleveland Clinic. “Aldosterone Antagonists.” ClevelandClinic.org. (Updated 2022 Apr 29; Accessed 2024 Mar 20).
- Shufelt, Chrisandra, and Alexis LeVee. “Hormonal contraception in women with hypertension.” JAMA 324.14 (2020): 1451-1452.
- Millán, Marta Martín, and Santos Castañeda. “Sex hormones and related compounds, including hormonal contraceptives.” Side Effects of Drugs Annual. Vol. 36. Elsevier, 2014. 615-634.
- FDA “Highlights of Prescribing Information: SLYND (drospirenone).” AccessData.FDA.gov (2019 May).